Qué
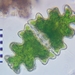
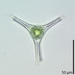
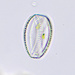
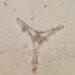
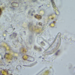
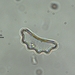
Género EuastrumObserv.
jennycallDescripción
second sampling site
about 130 um long and 78 um wide
pinnatum or oblongum
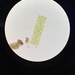
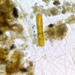
Fotos / Sonidos
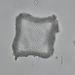
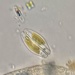
Observ.
vilseskogFecha
Septiembre 28, 2021Descripción
Freshwater pond, microscopy 2-60x, regular light and darkfield.
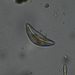
Fotos / Sonidos
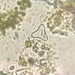
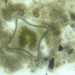
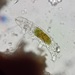
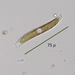
Observ.
tomleeturnerDescripción
51 microns across at thinnest point; 75 microns diagonally.
Fotos / Sonidos
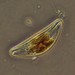
Qué
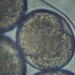
Género MilnesiumObserv.
curatorcarmineDescripción
terrestrial, from Candelaria concolor lichen
Fotos / Sonidos
Qué
Género GyrosigmaObserv.
jane_trembathDescripción
In freshwater with high conductivity, pH 8.5 and borderline eutrophic
Observ.
lamawebberDescripción
Eucampia zodiacus Ehrenberg 1839
A live image of the marine diatom Eucampia zodiacus Ehrenberg 1839. Class, Mediophyceae; Subclass, Chaetocerotophycidae; Family, Hemiaulaceae. E. zodiacus is the type species for this genus (Guiry & Guiry 2021).
Girdle view. Only five species of Eucampia reported (Round et al., 1990, Guiry & Guiry 2021). Cells often form flattened chains by interlocking the two short apical elevations (horns). The wide apertures between valves may be elliptical, oval, angular elliptical to square in outline. Valves are concave between the two elevations. The labiate process (rimoportulae) is centrally located on the valve. Helical-spiral chains form due to small differences in growth of the valves and girdle. Chloroplasts are round to ovoid bodies (Hendey 1964, Hasle & Syvertsen 1996, Hoppenrath 2009).
Eucampia zodiacus is a cosmopolitan marine centric diatom species, not reported from polar regions. Frequently found in plankton samples from Trincomali Channel, Galiano Island, BC., Canada (Waters 1972, Shim, 1976)
Methods:
From marine plankton collected in the Trincomali Channel, Spanish Hill Wharf, North Galiano Island, Gulf Islands, British Columbia, Canada, May 15, 2021. Live sample. Nikon TE300, 40x DIC NA 0.90.
References:
Guiry, M.D. & Guiry, G.M. 2012. AlgaeBase. World-wide electronic publication, National University of Ireland, Galway. http://www.algaebase.org; searched on 16 May 2021.
Hasle, G.R. & Syvertsen, E.E. (1996). Marine Diatoms. In: Identifying Marine Phytoplankton. (Tomas, C.R. Eds), pp. 173-176, Plate 35, p. 175. San Diego: Academic Press.
Hendey, N.I. (1964). An introductory account of the smaller algae of British coastal waters. Part V. Bacillariophyceae (diatoms). Her Majesty’s Stationary Office, London. pp. 107. Plate VII, fig. 1.
Hoppenrath, M., Elbrachter, M., Drebes, G. (2009) Marine Phytoplankton, Selected microphytoplankton species from the North Sea around Helgoland and Sylt. Schweizerbart’sche Verlagsbunchhandlung, Stuttgart, Germany. p. 98, Figs 42 j-q.
Round, F.E. Crawford, R.M. and Mann, D.G. (1990). The Diatoms, Biology & Morphology of the Genera, pp. 262-263. Cambridge University Press, Cambridge, UK.
Shim, J. H. (1976). Distribution and Taxonomy of Planktonic Marine Diatoms in the Strait of Georgia, B.C. Phd. Thesis, UBC. p. 152., p. 224 Plate II. Fig. 2.
Waters, R.E., Brown, L.N. and Robinson, M.G. (1972). Phytoplankton of Esquimalt Lagoon, British Columbia: Comparison with West Vancouver Island Coastal and Offshore Waters. Canadian Technical Report of Hydrography and Ocean Sciences. No. 37. Department of Fisheries and Oceans. p. 7, Table 2.
Observ.
vilseskogDescripción
In water sample from Delaware-Raritan Canal at Griggstown, with plankton net.
Fotos / Sonidos
Observ.
vilseskogDescripción
From water sample taken with plankton net in Delaware-Raritan Canal, Griggstown, NJ.
Fotos / Sonidos
Observ.
mnold1Descripción
Mag. 400x
For comparison see http://www.digicodes.info/Micrasterias_furcata.html#2014039005.
A pond edge water sample (freshwater) was taken on 9/10/2021 using a 10 micron dip net to enrich for microorganisms.
Fotos / Sonidos
Observ.
mnold1Descripción
Mag. 400x
For comparison see http://protist.i.hosei.ac.jp/PDB/Images/Chlorophyta/Micrasterias/mahabuleshwarensis.html.
A pond edge water sample (freshwater) was taken on 9/10/2021 using a 10 micron dip net to enrich for microorganisms.
Fotos / Sonidos
Qué
Género NaviculaObserv.
mnold1Descripción
Mag. 400x
A shoreline water sample was taken on 7/27/2021 from the Thames River estuary (New London county, CT - brackish) using a 10 micron dip net to enrich for microorganisms.
Fotos / Sonidos
Qué
Diatomeas (Clase Bacillariophyceae)Observ.
shanesmicroscopeDescripción
Collected 20210802 from a river tributary (Manatawny Creek, tributary of Schuylkill River), scoop of sediment near the bank. Added 1% milk that evening, then allowed to sit.
Fotos / Sonidos
Qué
Género AmphoraObserv.
shanesmicroscopeDescripción
Collected 20210802 from a river tributary (Manatawny Creek, tributary of Schuylkill River), scoop of sediment near the bank.
Fotos / Sonidos
Observ.
mnold1Descripción
Mag. 400x
A shoreline water sample was taken on 7/27/2021 from the Thames River estuary (New London county, CT - brackish) using a 10 micron dip net to enrich for microorganisms.
Qué
Diatomeas (Clase Bacillariophyceae)Observ.
jonathan_mackDescripción
found in algal mat in shallow man made pool/pond hybrid.
Qué
Diatomeas (Clase Bacillariophyceae)Observ.
jonathan_mackDescripción
found in algal mat in shallow man made pool/pond hybrid.
Fotos / Sonidos
Qué
Amphora ovalisObserv.
mnold1Descripción
Mag. 400x
Large Amphora (a little over 100µ), ventral view (I think). ID based on the large size and images here https://diatoms.org/species/amphora_ovalis.
A shoreline water sample was taken on 8/9/2021 from a cove off the Thames River estuary (New London county, CT - brackish) using a 10 micron dip net to enrich for microorganisms.
Fotos / Sonidos
Observ.
mnold1Descripción
Mag. 400x
See https://diatoms.org/genera/diploneis for comparison... this specimen looks more like D. elliptica (a touch of angularity) versus D. ovalis (smoothly curved over the entire perimeter). The size is in the range of both species.
A shoreline water sample was taken on 8/9/2021 from a cove off the Thames River estuary (New London county, CT - brackish) using a 10 micron dip net to enrich for microorganisms.
Fotos / Sonidos
Qué
Género GyrosigmaObserv.
shanesmicroscopeDescripción
Collected 20210802 from a river tributary (Manatawny Creek, tributary of Schuylkill River), scoop of sediment near the bank.
Fotos / Sonidos
Observ.
shanesmicroscopeDescripción
Collected 20210802 from a river tributary (Manatawny Creek, tributary of Schuylkill River), scoop of sediment near the bank.
Qué
Diatomeas (Clase Bacillariophyceae)Observ.
john_plunketDescripción
From HGTC retention pond. 400x.
Fotos / Sonidos
Qué
Género MelosiraObserv.
mnold1Descripción
Mag. 400x
3 Milosira were observed on this day, fitting the description: Large, Medium, and Small. This specimen was the largest form observed.
A shoreline water sample was taken on 7/27/2021 from the Thames River estuary (New London county, CT - brackish) using a 10 micron dip net to enrich for microorganisms.
Fotos / Sonidos
Qué
Género DiploneisObserv.
mnold1Descripción
Mag. 400x
A shoreline water sample was taken on 7/27/2021 from the Thames River estuary (New London county, CT - brackish) using a 10 micron dip net to enrich for microorganisms.
Fotos / Sonidos
Observ.
mnold1Descripción
Mag. 400x
A shoreline water sample was taken on 8/9/2021 from a cove off the Thames River estuary (New London county, CT - brackish) using a 10 micron dip net to enrich for microorganisms.
Fotos / Sonidos
Qué
Género NitzschiaObserv.
mnold1Descripción
Mag. 400x
Not certain of the ID. Seems shorter and wider than images I can Google. Also more tapered at the apices. Might this be a daughter cell that has gone through many divisions thereby acquiring a shortened frustule? (if this is indeed N. sigmoidea) - For Googled images, see: https://www.algaebase.org/search/images/detail/?img_id=33818 and http://nordicmicroalgae.org/taxon/Nitzschia%20sigmoidea. AglaeBase specifies this organism as a freshwater specie.
A shoreline water sample was taken on 7/27/2021 from the Thames River estuary (New London county, CT - brackish) using a 10 micron dip net to enrich for microorganisms.
Fotos / Sonidos
Qué
Género GyrosigmaObserv.
shanesmicroscopeDescripción
Collected 20210802 from a river tributary (Manatawny Creek, tributary of Schuylkill River), scoop of sediment near the bank. Added 1% milk that evening, then allowed to sit.
Fotos / Sonidos
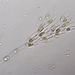
Qué
Osos de Agua (Filo Tardigrada)Observ.
animalculeDescripción
tardigrade eggs from a large sample i'm keeping as a self-sustaining microbiome in a chinese takeout container! not sure the exact species but i have them on a slide in a humidity chamber so i can keep my eye on them! (i actually caught the mom while pregnant, she laid the eggs while on the slide! i named her mocha, so all the babies will have choco names, like java chip, choco curl, malt...)
Fotos / Sonidos
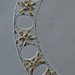
Qué
Género DinobryonObserv.
mnold1Descripción
Sample collected (3/9/2020) at water's edge using a stick to extract filamentous algae and depositing the algae in a small plastic vial containing water from the same location. Air temp. 56F